Metal–Organic Frameworks (MOFs): Gas Separation and Mechanochemical Synthesis
Porous Materials Chemistry · CO₂ Capture · Green Synthesis`
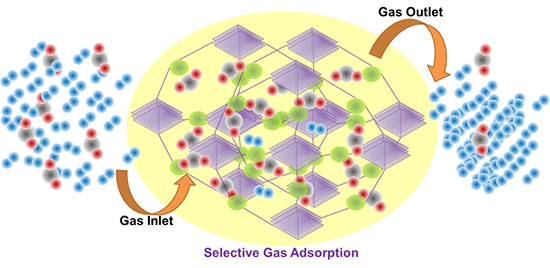
Metal–organic frameworks (MOFs) are among the most structurally versatile porous materials ever developed — recognized by IUPAC as a top emerging technology — with precisely tailorable pore geometries and exceptional surface areas that make them powerful candidates for selective gas separation and carbon capture. Our MOF program pursues two interconnected threads.
In collaboration with Dr. Yang Qin (Department of Chemistry & Chemical Biology, University of New Mexico), we investigate charge-separated MOFs for highly selective CO₂ capture. Conventional MOFs require charge-balancing guest ions that occupy pore space and limit capacity. Our charge-separated design eliminates free ions entirely by pairing anionic and cationic framework components, yielding cleaner, more accessible pores. Our benchmark material, UNM-1 (C₅₂H₁₆BCuF₁₆N₄), pairs an anionic tetrahedral borate ligand with a cationic Cu(I) center to form a diamondoid network with four-fold interpenetration — simultaneously delivering exceptional environmental stability and a BET surface area of 621 m² g⁻¹. Gas adsorption measurements reveal temperature-dependent CO₂ adsorption/desorption hysteresis and CO₂/N₂ ideal selectivities reaching ~99 at 313 K and 1 bar, highlighting the promise of charge-separated MOFs for flue gas treatment and post-combustion CO₂ sequestration.
In a separate but complementary collaboration with Dr. Wen-Yang Gao, we develop mechanochemical — solvent-free, low-energy — synthetic routes to structurally novel MOFs. Our work has established systematic mechanochemical access to Group 4 element-based MOFs (Zr, Hf), including cluster-precise multimetallic frameworks that cannot be accessed by conventional solvothermal synthesis, and has recently extended to Pd(II)-based heterometallic lattice sites with potential catalytic applications. In our laboratory, we characterize all materials using adsorption isotherms, spectroscopic methods, and surface analysis to connect synthetic strategy to functional performance.

ADMINISTRATIVE ASSISTANT
GAYAN R. RUBASINGHEGE
Associate Professor of Chemistry
New Mexico Institute of Mining and Technology
Department of Chemistry
801 Leroy Place
Socorro, NM 87801
Bethany Jessen
New Mexico Institute of Mining and Technology
Department of Chemistry
801 Leroy Place
Socorro, NM 87801
Phone: 575-835-5129
Fax: 575-835-5215
Phone: 575-835-5263
Fax: 575-835-5364
Copyright © 2018 The Environmental Chemistry Research Research Group. All rights reserved.